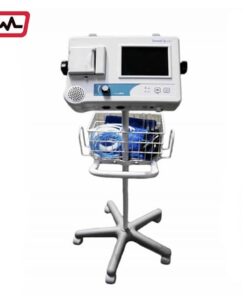
Summit Doppler VANABI Vantage ABI
NEW
Patient Ready
Warranty Included

Category: DOPPLER
Tags: ABI System, Doppler, Summit AVS ABi system, Summit doppler, Vascular Doppler, Vascular System
The Vantage ABI uses a special new cuff-based technology for performing the ankle-brachial index (ABI) exam to make the diagnosis of peripheral arterial disease (P.A.D.) simple. The system provides an accurate determination of the ABI across the full range of P.A.D. This technology, developed by Summit Doppler, was co-invented in cooperation with researchers from the well known Cleveland Clinic. It uses DFO waveform analysis, which is a significant improvement in ankle pressure determination and peripheral arterial disease assessment over traditional forms of oscillometric ankle pressure estimation.
Package 1 (VANABI) includes:
- Main Unit with Easy to Use Touch Screen
- PC Download Software
- USB Stick – for easy storage of exams
- 4 Cuffs and Hoses
Dimensions:
- Depth: 11 in/27.9 cm
- Width: 7.3 in/18.5 cm
- Height: 6.4 in/16.25 cm
- Weight: 4.6 lbs/2.1 Kg
- Mounting: Self‐supported (rubber feet) or stand mounted (optional)
- Portability: Built‐in carry handle
Clinical: - Cuff Pressure Range: 0 to 280 mmHg
- Pressure Gage Accuracy: +/-3 mmHg 0‐200 mmHg, +/-2% >200 mmHg
- Deflation Rate: 2.5 mmHg/second nominal
- Inflation Pressure: 180 mmHg (Default); 280 mmHg (User selected)
- Deflation Pressure: 60 mmHg (Default); 35 mmHg (User selected)
- Pulse Rate Range: 43 to 120 BPM
- ABI Determination Time: 3 minutes
- Determination: Pulse Volume Oscillometry – Patent Pending
- Waveform Type: Pnuematic Plethysmography ( Pulse Volume Recording ‐PVR)
- Patient Connections: 4 Vascular Cuffs, 6 foot hose length (ea.)
- Clinical Sensitivity: > 90%
- Clinical Specificity: > 92%
Battery: - Type: 7.2V NiMH (custom pack with thermal and over‐current protection)
- Replacement Part: B200
- Replacement Interval: 1 year
External Power Adaptor: - Input: 100-240VAC 50/60 Hz 1.2 Amp
- Output: + 7VDC, 5A
Environmental: - Operating Temperature: 10 – 30° C
- Operating Humidity: 30 – 75% non‐condensing
- Transport/Storage Temperature: [-20] to 50° C
- Transport/Storage Humidity: 5 – 90% non‐condensing